
Indiva Reports Third Quarter 2022 Results
Indiva Launches Multiple New Products and Remains the National Market Share Leader in the Edibles Category
LONDON, Ontario – November 22, 2022: Indiva Limited (the “Company” or “Indiva”) (TSXV:NDVA) (OTCQX:NDVAF), the leading Canadian producer of cannabis edibles and other cannabis products, is pleased to announce its financial and operating results for the third fiscal quarter ended September 30, 2022. All figures are reported in Canadian dollars ($), unless otherwise indicated. Indiva’s financial statements are prepared in accordance with International Financial Reporting Standards (“IFRS”). For a more comprehensive overview of the corporate and financial highlights presented in this news release, please refer to Indiva’s Management’s Discussion and Analysis of Financial Condition and Results of Operations for the Three and Nine Months Ended September 30, 2022, and the Company’s Condensed Consolidated Interim Financial Statements for the Three and Nine Months Ended September 30, 2022 and 2021, filed on SEDAR and available on the Company’s website, www.indiva.com.
“We are pleased to report year-over-year growth and record net revenue year-to-date, driven by the successful launch of several new products and brands into the Canadian market in Q3 2022, including Indiva Life Double Stuffed Sandwich Cookies, Indiva Life Lozenges, Dime vapes and Pearls by Grön gummies, which have quickly become one of the top edibles brands in market. Indiva is transitioning from relying on licensed brands, which Indiva will continue to support with its best efforts, towards focusing on innovation at its core, in order to drive future growth,” said Niel Marotta, President and Chief Executive Officer of Indiva. “In the third quarter, Indiva launched a record number of SKUs, primarily into the province of Ontario and B.C. These new products are now beginning to roll out nationally across our distribution platform, which spans all thirteen provinces and territories in Canada. Feedback from key accounts and budtenders has been very positive, which has translated into robust replenishment orders for our new products. We look forward to continuing to delight of-age Canadian cannabis enthusiasts with the quality and innovation that Indiva products are known for.”
Quarterly Performance
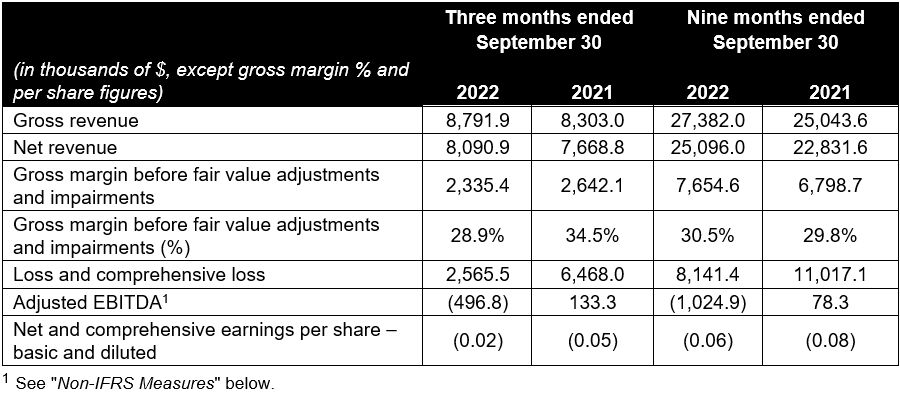
- Gross revenue in Q3 2022 was $8.8 million, representing a 1.1% sequential decrease from Q2 2022, and a 5.9% increase year-over-year from Q3 2021. Year-to-date, gross revenue increased 9.3% year over year to a record $27.4 million.
- Net revenue in Q3 2022 was $8.1 million, representing a 0.4% sequential decrease from Q2 2022, and a 5.5% increase year-over-year from Q3 2021, driven by new product introductions and strength in core products. Q3 2022 net revenue was negatively impacted by the closure of the B.C. Liquor Distribution Branch distribution center due to strike action, as well as the partial shutdown of the Ontario Cannabis Retail Corporation, operating as the Ontario Cannabis Store, (the “OCS”) distribution center due to a cyber security incident, resulting in delays in deliveries and new product launches by up to two weeks. Year-to-date, net revenue increased 9.9% year over year to a record $25.1 million.
- Net revenue from edible products in the quarter was $7.3 million, up slightly from $7.2 million in Q2 2022 and $6.9 million in the prior year period. Edible product sales represent 90.7% of net revenue in Q3 2022. Year-to-date net revenue from edible products increased 10.7% year-over-year to a record $23.1 million or 92.0% of net revenue.
- Gross profit before fair value adjustments, impairments and one-time items declined year-over-year and sequentially, to $2.3 million, or 28.9% of net revenue, versus 33.1% in Q2 2022 and 34.5% in Q3 2021. The decline in gross margin was due to the delay in receiving automation equipment related to the production and processing of certain new products, offset by lower inventory write-downs. Year-to-date, gross profit before fair value adjustments, impairments and one-time items increased to a record $7.7 million, or 30.5% of net revenue, versus $6.8 million or 29.8% of net revenue in the corresponding period last year.
- In Q3 2022, Indiva sold products containing 56.5 million milligrams of cannabinoids, the active ingredient in edible products, which represents a 32.4% increase when compared to the 42.7 million milligrams in products sold in Q2 2022, and a 33.7% increase compared to 42.3 million milligrams sold in Q3 2021. The increase was primarily driven by a shift in product mix towards edibles with higher total cannabinoid content and the addition of minor cannabinoids (ie. CBN and CBG) to multiple SKUs.
- Impairment charges in the quarter totaled $0.4 million. This impairment includes a write off of aged finished goods and bulk cannabis flower, and to a lesser extent, certain packaging for obsolete products, offset by a recovery on oil-based products. The Company will continue to work to monetize any impaired inventory which remains saleable. The Company expects lower inventory impairments going forward as most of the bulk flower inventory originating from terminated contract manufacturing has either been sold or written down to its net realizable value.
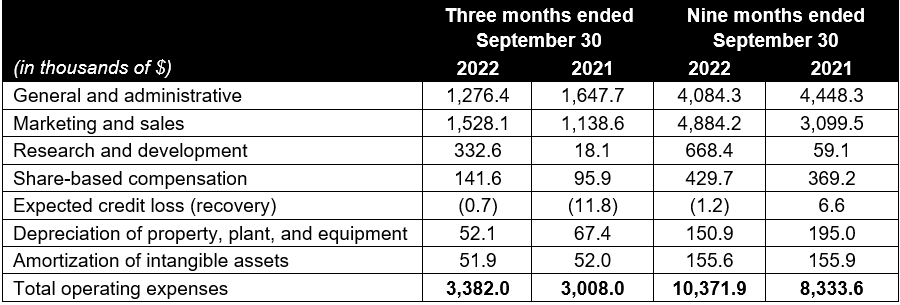
- Operating expenses in the quarter decreased 3.0% sequentially, representing 41.8% of net revenue, versus 42.9% in Q2 2022 and 39.2% in Q3 2021. Operating expenses declined due to lower general and administrative costs, which were down 22.5% year-over-year and down 6.1% sequentially, offset by higher marketing costs and sales commissions versus the prior year. Year-to-date, operating expenses increased by 24.5% to $10.4 million due to higher marketing costs and sales commissions, higher research and development costs as a result of new product innovation activities, offset partly by lower general and administrative expenses.
- Adjusted EBITDA declined sequentially in Q3 2022 to a loss of $0.5 million, versus a loss of $0.15 million in Q2 2022, and declined versus a profit of $0.13 million in Q3 2021, due to higher cost of goods related to new product launches, increased sales and marketing costs year-to-date and research and development expenses, offset partly by lower general and administrative costs. Year-to-date, adjusted EBITDA was a loss of $1.0 million versus a profit of $0.1 million in the corresponding period last year. See “Non-IFRS Measures” below.
- Comprehensive net loss of $2.6 million included one-time expenses and non-cash charges for impairment of inventory totaling $0.4 million. Excluding these charges, comprehensive loss increased to $2.2 million versus an adjusted loss of $2.0 million in Q2 2022 and $1.0 million in Q3 2021.
- Cash balance improved to $3.6 million at quarter-end.
Operational Highlights for the Third Quarter 2022
- Indiva completed initial deliveries of Pearls by Grön gummies to Ontario, B.C., Manitoba and Saskatchewan. The first four flavours released include Blue Razzleberry 3:1 CBG:THC, Sour Apple THC, Blackberry Lemonade 1:1:1 CBN:CBD:THC and Pomegranate 4:1 CBD:THC. Pearls by Grön have quickly become one of the top-selling edibles in Ontario.
- Towards the very end of the third quarter, Indiva introduced several additional new products to provincial wholesalers including:
- Pearls by Grön: Strawberry Melon 4:1 CBN:THC, Sparkling Peach 1:1: CBD:THC and Cherry Limeade THC, bringing the total number of Pearls gummie SKUs in market to seven. These additional flavours were delivered to the OCS in late September and became available to retailers in October.
- Indiva Life Double Stuffed Sandwich Cookies: Available in Vanilla and Fudge flavours, containing 10mg of THC per cookie, these products became available in Ontario, Alberta, B.C. and New Brunswick in Q3, adding substantially to Indiva’s market share in the baked goods sub-category. The Company expects to expand distribution of this immediately popular product across the country in the fourth quarter.
- Indiva Life Capsules: Three new 30-count capsule formats became available in the third quarter, including Zen CBD:CBN 1:1, Sunrise CBG:THC 1:1 and Sunset CBN:THC 1:1. Indiva Life Zen capsules became available in Alberta, while Zen, Sunrise and Sunset capsules became available in B.C., and Sunset and Zen capsules became available in Manitoba, Saskatchewan and the Yukon.
- Indiva Life Lozenges: This innovative extract product comes in Lemon and Wild Cherry flavours, available in 10-pack and 25-pack counts. Lemon Lozenges became available in Ontario in a 10-pack format, and in B.C. in a 25-pack format during the quarter. 25-pack Wild Cherry Lozenges were delivered to Ontario and B.C. subsequent to quarter end.
- Indiva Life Chocolates: Irish White Chocolate THC delivered in Ontario, with Evening Milk Chocolate CBN:CBD 1:1 and Afternoon Trail Mix Milk Chocolate CBG:THC 1:1 becoming available subsequent to quarter end. Evening Milk and Afternoon Trail Mix delivered to B.C. in October.
- Bhang Chocolate: Toffee and Salt Milk Chocolate and White Chocolate Candy Cane became available in Ontario.
- Dime Industries Vapes: 510-thread Dime OG carts delivered in Ontario. Indiva also introduced a proprietary Dime battery and 510-thread Bubblegum Kush carts, both of which became available in October.
Events Subsequent to Quarter End
- Indiva was awarded thirteen additional SKU listings by the OCS, which will deliver to the OCS late December/early January. New listings include:
- Pearls by Grön: Marionberry Lemonade CBG 25-pack gummies.
- Wana: Midnight Berry Indica CBN/CBD/THC 5:10:2 available in 5-packs.
- Indiva Life Double Stuffed Sandwich Cookies: Golden Vanilla and Be My Valentine Strawberry flavours, each cookie containing 10mg of THC.
- Indiva Life Capsules and Lozenges: Sunrise CBG/THC 1:1 capsules, as well as Lemon Lozenges available in 25-count pack and Wild Cherry Lozenges in 50-count packs.
- Indiva Life Chocolates: Morning Espresso Milk Chocolate 1:1 CBG:THC and Raspberries and Cream White Chocolate 1:1 CBD:THC.
- Dime Industries Vapes: 510-thread Blueberry Lemon Haze Sativa and Wedding Cake Hybrid Rechargeable All-in-One.
- Building on Indiva’s strength as a best-in-class manufacturer, the Company commissioned several new pieces of automated equipment at its facility in London, Ontario, for use in the processing and packaging of edible products. The Company expects that by year-end, all of the new equipment, including pieces specifically designed for use in the processing of newly launched products, will be fully operational, adding finished goods capacity, while lowering operating costs. Despite the reduction in required direct labour for certain processing and packaging activities, the addition of several new brands and products has created a growth environment at Indiva’s production facility, requiring additional direct labour requirements, thus preventing any job cuts.
Market Share
- Data from Hifyre Inc. for the third quarter of 2022 shows strong sell-through of Indiva edible products. With 29.7% share of sales, Indiva continues to lead in the #1 market share position in the edibles category on an aggregate basis. Please note that Hifyre data for the three-month period ended September 30, 2022 does not include any contribution from Pearls by Grön gummies.
- Ontario: #1 with 28.2% market share.
- Alberta: #1 with 28.1% market share.
- British Columbia: #1 with 37.4% market share.
- Saskatchewan: #2 with 17.8% market share.
- Manitoba: #2 with 32.1% market share.
- Wana™ Sour Gummies led the edibles category with 24.7% category share and 31.9% sub-category share, and Bhang® continued to lead the chocolate category with 37.2% sub-category share.
- Product ranking in Q3 2022 showed four of the Top 10 edible SKUs are from Indiva.
- Based on data from British Columbia, Alberta, Ontario, Manitoba and Saskatchewan, the edibles category declined by 1.0% in Q3 2022 to $57.0 million in retail sales from $57.6 million in Q2 2022.
Outlook
- Based on the strength of purchase orders received to date in the fourth quarter, the Company expects Q4 2022 net revenue to be higher sequentially and year-over-year driven primarily by new product introductions, including Pearls gummies, Dime Industries vape products, as well as new Indiva Life branded products such as lozenges and sandwich cookies.
- Margins are expected to improve going forward due to fixed cost leverage and the commissioning and implementation of automation in the production, processing and packaging of edible products.
Operating and Financial Results for the Three and Nine Months ended September 30, 2022 and 2021
Operating Expenses
CONFERENCE CALL – Tuesday, November 22, 2022 at 8:30 a.m. (EST):
The Company will host a conference call to discuss its results on Tuesday, November 22, 2022 at 8:30 a.m. (EST). Interested participants can join by dialing 416-764-8658 or 1-888-886-7786. The conference ID is 70018162.
A recording of the conference call will be available for replay following the call. To access the recording please dial 416-764-8691 or 1-877-674-6060. The replay ID is 018162#. The recording will remain available until Thursday, December 22, 2022.
About Indiva
Indiva sets the standard for quality and innovation in cannabis. As a Canadian licensed producer, Indiva produces and distributes award-winning cannabis products nationally, including Bhang® Chocolate, Wana™ Sour Gummies, Jewels Chewable Tablets, Grön edibles, Dime Industries™ vape products, as well as capsules, edibles, extracts, pre-rolls and premium flower under the INDIVA, Indiva Life and Artisan Batch brands. Click here to connect with Indiva on LinkedIn, Instagram, Twitter and Facebook, and here to find more information on the Company and its products.
Contacts
Investor Contact
Anthony Simone
Phone: 416-881-5154
Email: [email protected]
Disclaimer and Reader Advisory
General
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) has in any way passed upon the merits of the contents of this news release and neither of the foregoing entities accepts responsibility for the adequacy or accuracy of this news release or has in any way approved or disapproved of the contents of this news release.
Certain statements contained in this news release constitute forward-looking information. These statements relate to future events or future performance. The use of any of the words “could”, “intend”, “expect”, “believe”, “will”, “projected”, “estimated” and similar expressions and statements relating to matters that are not historical facts are intended to identify forward-looking information and are based on the parties’ current belief or assumptions as to the outcome and timing of such future events. Actual future results may differ materially. In particular, this news release contains forward-looking information relating to, among other things, (i) the Company’s outlook for and expected operating margins and future financial results, (ii) the projected growth of its business and operations (including existing and new segments thereof), and the future business activities of, and developments related to, the Company within such segments after the date of this news release, including the anticipated introduction of new product offerings (iii) the Company’s ability to capture and/or maintain its market share in any jurisdiction, and (iv) the Company’s ability to deliver on its commitments for existing or new listings of products. Various assumptions or factors are typically applied in drawing conclusions or making the forecasts or projections set out in forward-looking information. Those assumptions and factors are based on information currently available to the Company, and include, without limitation, assumptions about the Company’s future business objectives, goals, and capabilities, the cannabis market, the regulatory framework applicable to the Company and its operations, and the Company’s financial resources. Although the Company believes that the assumptions underlying, and the expectations reflected in, forward-looking statements in this news release are reasonable, it can give no assurance that such expectations will prove to have been correct. A number of factors could cause actual events, performance or results to differ materially from what is projected in the forward-looking statements. Specifically, readers are cautioned that forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company, as applicable, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements, including, but not limited to, risks and uncertainties related to: (i) the available funds of the Company and the anticipated use of such funds, (ii) the availability of financing opportunities, (iii) legal and regulatory risks inherent in the cannabis industry, (iv) risks associated with economic conditions, (v) dependence on management, (vi) public opinion and perception of the cannabis industry, (vii) risks related to contracts with third-party service providers, (viii) risks related to the enforceability of contracts, (ix) reliance on the expertise and judgment of senior management of the Company, and ability to retain such senior management, (x) risks related to proprietary intellectual property and potential infringement by third-parties, (xi) risks relating to the management of growth and/or increasing competition in the industry, (xii) risks associated to cannabis products manufactured for human consumption, including potential product recalls, (xiii) risks related to the economy generally, and (xiv) risk of litigation.
The forward-looking information contained in this news release is made as of the date hereof and the Company is not obligated to, and does not undertake to, update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as required by applicable securities laws. Because of the risks, uncertainties and assumptions inherent in forward-looking information, investors should not place undue reliance on forward looking information. The foregoing statements expressly qualify any forward-looking information contained herein.
This news release contains future-oriented financial information and financial outlook information (collectively, “FOFI“) about the Company’s prospective results of operations, which are subject to the same assumptions, risk factors, limitations, and qualifications as set out in the above paragraph. FOFI contained in this news release was approved by management as of the date of this news release and was provided for the purpose of providing further information about the Company’s future business operations. The Company disclaims any intention or obligation to update or revise any FOFI contained in this news release, whether as a result of new information, future events or otherwise, unless required pursuant to applicable law. Readers are cautioned that the FOFI contained in this document should not be used for purposes other than for which it is disclosed herein.
Non-IFRS Measures
This news release makes reference to certain non-IFRS measures. These measures are not recognized measures under IFRS, do not have a standardized meaning prescribed by IFRS, and are therefore unlikely to be comparable to similar measures presented by other companies. Rather, these measures are provided as additional information to complement those IFRS measures by providing further understanding of our results of operations from management’s perspective. Accordingly, these measures should not be considered in isolation nor as a substitute for analysis of our financial information reported under IFRS.
The non-IFRS measure used in this news release includes “Adjusted EBITDA”. The Company calculates Adjusted EBITDA as a sum of net revenue, other income, cost of inventory sold, production salaries and wages, production supplies and expense, general and administrative expense, and sales and marketing expense, as determined by management. Adjusted license fee eliminates 50% of the fee which is equivalent to the Company’s share of the joint venture company to which the license fee is paid. Adjusted EBITDA is provided to assist readers in determining the ability of the Company to generate cash from operations and to cover financial charges. Management believes that Adjusted EBITDA provides useful information to investors as it is an important indicator of an issuer’s ability to generate liquidity through cash flow from operating activities and equity accounted investees. Adjusted EBITDA is also used by investors and analysts for assessing financial performance and for the purpose of valuing an issuer, including calculating financial and leverage ratios. The most directly comparable financial measure that is disclosed in the financial statements of the Company to which the Non-IFRS measure relates is income (loss) from operations.